Comprehensive
Across clinical, genetics, experimental and text sources.
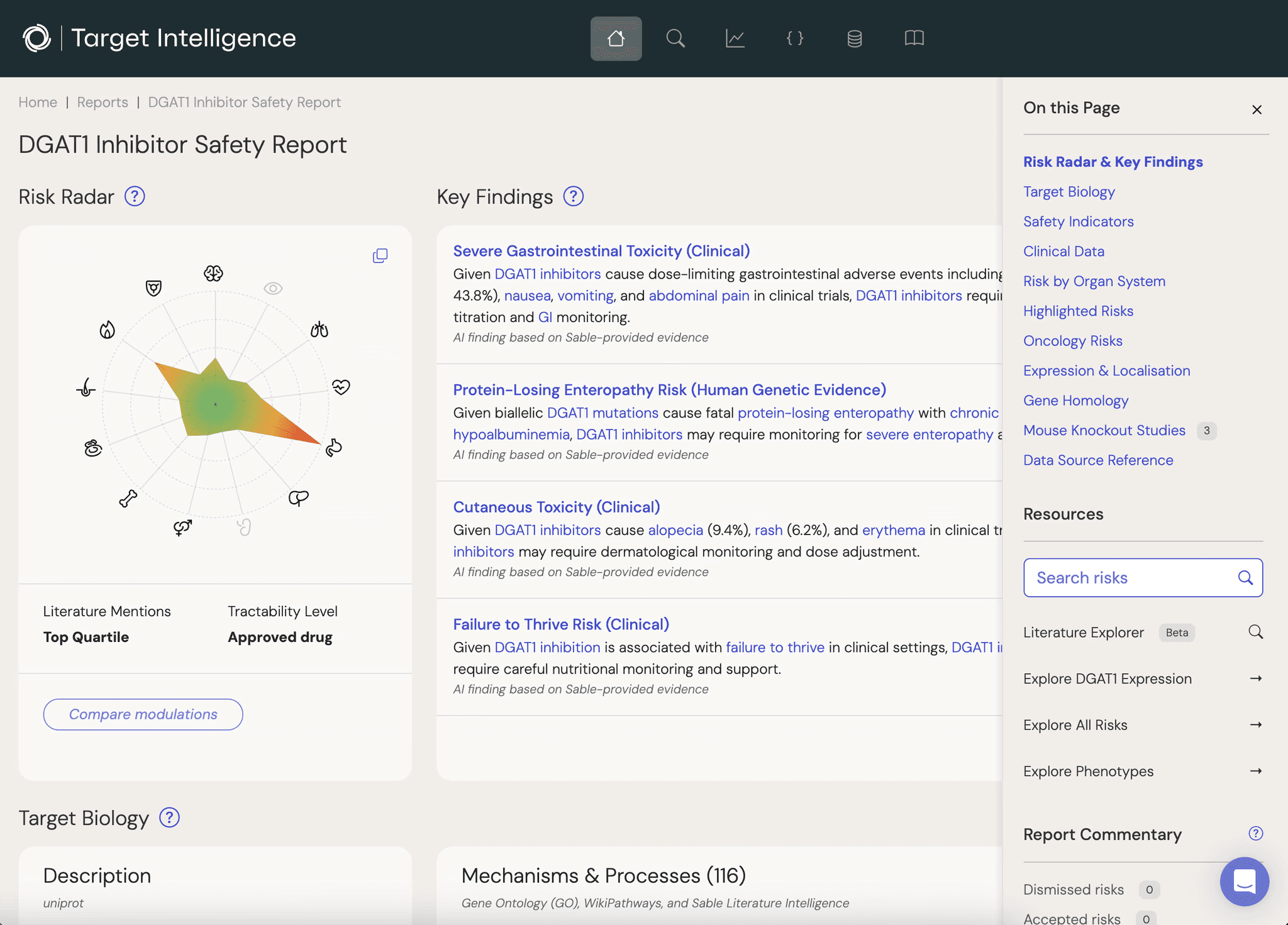
Safety Specific
Digest relevant risks without getting lost in endless data.
Interactive
Reports respond to your chosen modality and preferences.
Fresh
Return to fresh data and regular improvements.
Features
Sable Target Intelligence is built to make target safety assessment more systematic, quantified, and convenient for toxicologists and scientists working across drug discovery.
Multi-modal Safety Data
Unite multiple safety-relevant perspectives in one place, covering genomic, experimental, animal model, scientific literature, clinical trial and other sources.
Data is organised by the direction of effect, ensuring theoretical risks align with your proposed mode of action (inhibition or activation).
Quickly build reports that interrogate our database of millions of theoretical risks, providing a systematic and prioritised review of safety areas for your selected drug target.
Convenient Risk Assessment and Collaboration
Theoretical risks are aggregated and weighted by severity and score, making comparisons straightforward.
Easily navigate from high-level summary to detailed evidence: explore key context, assess individual relationships, and verify references, all within one place.
Leverage your expertise to assess risks: dismiss, comment, and collaborate with your team using a structured commenting system.
Use cases
These are the use-cases we work on with our customers:
Target Safety Assessment
Evaluate theoretical safety risks associated with a selected target during drug discovery.
Bulk Safety Assessment
Compare safety across multiple targets where individual manual assessment is impractical.
Target Due Diligence
Anticipate potential risks for a target in development by a third party.
What scientists say
Here's what scientists have said about Sable Target Intelligence.